By Allen Chiu and Certina Ho
While the intrinsic nature of healthcare is to do good, the opposite may unintentionally occur. For example, something as simple as combining two pills as one in order to provide convenience and improve adherence in patients can still have the potential to cause patient harm. The following medication incident was anonymously reported to the Institute for Safe Medication Practices Canada (ISMP Canada):
“Patient was admitted to emergency with confusion and hypotension. It was later found out that the patient took both metformin 1000 mg twice daily and a combination metformin/sitagliptin 1000/50 mg tablet twice daily. Several weeks ago, it was suggested that the patient switched to a combination pill [metformin/sitagliptin 1000/50 mg], but the old medication [metformin 1000 mg] was not discontinued.”*
*Note: The maximum dose of metformin is 2550 mg per day. In this case, the patient has taken a total of metformin 4000 mg per day due to duplicate therapy.
Combination drugs or fixed-dose combinations, for the purpose of this article, refer to a medication that contains more than one active ingredient. They are also sometimes referred to as “combo pills” or “polypills”. Using combination drug products can potentially reduce pill burden and may increase patient adherence, leading to better patient outcomes. This strategy may also reduce the financial cost to the patient. Fixed-dose combinations are most commonly seen in anti-hypertensives and anti-hyperglycemics, but they can also be found in other therapeutic categories, such as, glaucoma eye drops and anti-retrovirals, etc. A 2015 report of prescription data by the Canadian Healthcare Network showed that combination medications were the top two most commonly prescribed within the therapeutic class of cardiovascular drugs. Table 1 provides some examples of these combination drug products.
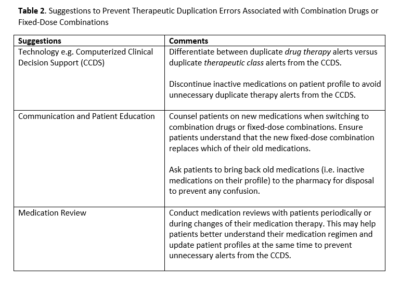
However, as seen in the above medication incident, using combination drugs can also inadvertently cause patient harm. This may not be an isolated case. A 2018 study on Irish prescription claims reported that therapeutic duplication errors were more likely in anti-hypertensive fixed-dose combinations compared to their individual components. While the above incident involved a different class of medications (i.e. anti-hyperglycemics), it was a therapeutic duplication error of metformin. While more research may be warranted on this topic, Table 2 offers some suggestions that may potentially decrease the risk of these incidents. In conclusion, while switching patients from single to combination drugs can have many benefits, healthcare practitioners should be aware of the potential risks associated as well.
Table 1. Examples of Combination Drugs or Fixed-Dose Combinations (Note: This is not a comprehensive list.)
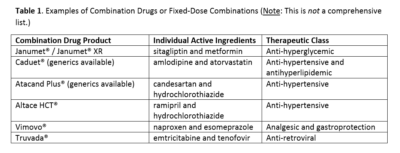
Table 2. Suggestions to Prevent Therapeutic Duplication Errors Associated with Combination Drugs or Fixed-Dose Combinations
Allen Chiu is a PharmD Student at the Leslie Dan Faculty of Pharmacy, University of Toronto; and Certina Ho is a Project Lead at the Institute for Safe Medication Practices Canada (ISMP Canada).