HN Summary
• Hamilton Health Sciences is the first hospital in North America to trial tiny carotid artery filters designed to trap blood clots and prevent strokes in high-risk patients.
• The global Phase 3 study will assess whether adding filters to standard blood thinner treatment can reduce stroke rates and improve outcomes.
• Early results show the minimally invasive, same-day procedure is quick, safe, and promising, highlighting innovation in stroke prevention.
Filters keep coffee fresh, water clean, and cars running smoothly.
Now, a team of Hamilton Health Sciences (HHS) doctors and researchers are using that same concept to prevent strokes, by placing tiny filters in patients’ carotid arteries at each side of the neck to trap blood clots so they can’t reach the brain.
HHS is the first hospital site in North America and most of the world to perform this innovative new procedure to investigate whether these tiny filters, called Vine carotid filters, can lower the number of strokes caused by large vessel blockages inside the brain.
This global, Phase 3 clinical trial is taking place through a partnership with a medical device company and our Hamilton General Hospital’s stroke and neuro-intervention teams. The trial is also exploring whether these filters will reduce the overall number of strokes, and will further evaluate the filters’ safety.
The clinical trials
Clinical trials are studies that evaluate promising new treatments not yet in regular use. They can be an option when standard treatments aren’t working well or aren’t the best choice for a patient.
Trials happen in phases so researchers can study their effectiveness in a gradual and safe way. Phase 1 trials explore tolerability and dosing, Phase 2 trials evaluate how well an intervention works and what side effects may occur; Phase 3 look at efficacy; and Phase 4 focuses on what happens in the long-term.
The Phase 1 and 2 trials took place in Europe, where close to 100 patients received filters. The Hamilton-led Phase 3 trial is much larger. Dr. Ashkan Shoamanesh, an HHS neurologist, and Dr. Alexander Benz, a cardiologist fellow at HHS, are leading the Phase 3 study globally, which will involve 2,000 patients worldwide over the next two to three years, with about 40 from HHS.
With Phase 3, one group of patients receives the filters along with the current, standard treatment of blood thinners, while other group continues with blood thinners only, and their outcomes are compared.
HHS has enrolled three patients so far. One underwent the procedure while the other two continued with the standard treatment of blood thinners.
The HHS patient who received the filters recovered quickly and is doing very well, says HHS neurologist Dr. Aristeidis Katsanos, the study’s site lead.
Katsanos, Shoamanesh and Benz all conduct research through the Population Health Research Institute, a world-renowned, shared institute of HHS and our academic partner McMaster University.
A promising innovation
Strokes in people with a history of atrial fibrillation (AF), an irregular heart rhythm, are often caused by blood clots that travel to the brain and block major blood vessels, which can lead to severe, life-altering disabilities. Since blood thinners don’t prevent all strokes in these patients, researchers are looking for other, more effective ways to protect them in addition to standard- of-care blood thinners.
Laboratory testing shows that these filters can trap larger clots (about 1.4 millimetres in size or bigger) and sometimes smaller ones too.
In people with AF, blood clots can form in the heart and travel through the bloodstream where they can cause a stroke if they reach the brain. Placing these filters in both carotid arteries, the main vessels carrying blood to the front of the brain, could stop large clots from the heart from reaching this area, where about 90 per cent of AF-related strokes occur.
Patients qualifying for this Phase 3 trial have had a stroke within the last year. They also have AF, which puts them at higher risk of stroke. While blood thinners help lower the risk, people with AF who have had a stroke in the past year face about a three to seven per cent chance of having another one.
Same-day procedure
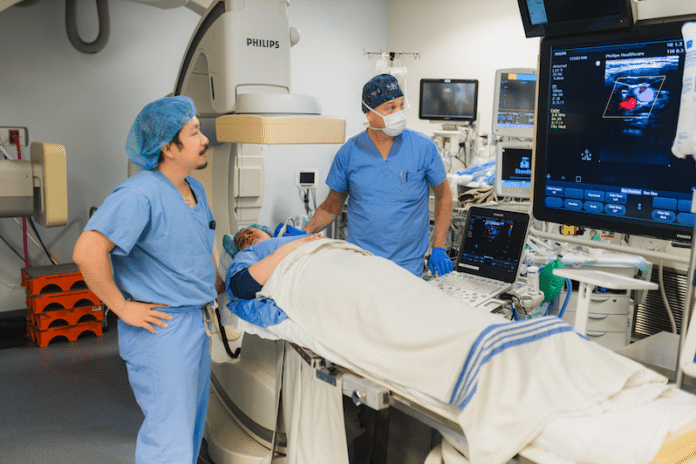
The HHS procedure was performed by neurointerventionalist and neurologist Dr. Brian Van Adel and neurosurgeon Dr. Bill Wang. A neurointerventionalist fixes blood vessel problems through small, precise procedures inside the body rather than open surgery.
The same-day procedure took just a few minutes and didn’t require sedation. The doctors injected a hair-thin nickel wire directly into the patient’s carotid artery under ultrasound guidance. Inside the carotid artery, the wire forms into a filter-like coil for catching clots so they can’t continue on to the brain.
“Researchers believe these filters could greatly reduce strokes caused by large blockages in these arteries, which are often the most severe and disabling,” says Katsanos, adding,
“This Phase 3 trial speaks to HHS’ expertise in stroke care and research globally, and is an excellent example of how teamwork across specialties is helping push stroke care forward, for better patient outcomes.”
Testing tomorrow’s treatments
All HHS patients are automatically enrolled in Explore Research, the hospital’s patient recruitment process for research studies. Patients are notified about Explore Research during their hospital visit and can opt out at any time if they’d prefer not to be contacted about opportunities to participate in research studies.